
Organic Chemistry in "Real Life"
This semester we will talk a lot about synthesis. Synthesis involves the conversion of one molecule into another that is usually larger and more complex, by performing a series of chemical reactions and transformations. As we mentioned in class, figuring out how to synthesize a molecule from a provided simpler starting material is called retrosynthetic analysis.
Synthesis of organic molecules is one of the main tasks of organic chemists. Believe it or not, there really is a joy in watching crystals grow of a molecule that nobody has ever made before. You hold the only example of that particular organic structure that exists in the world! Unfortunately, I am a very poor synthetic organic chemist, and I have experienced this only rarely!!
Organic synthesis may be said to have started around the turn of the 19th century in Germany, where chemists sought ways to make dyes other than those obtained from plants. Dyes were very important commercially at the time, and remain so to this day. An obvious example for me is in the area of photography (I used to work at Eastman Kodak). Chemists at Kodak have synthesized over 30,000 organic dyes for various uses in photographic film paper, filters etc. Dyes are also synthesized, for example, to satisfy the desire of daughter to change her hair to any color she likes! This blue was from a few years ago, by now she has dyed her hair so many different times that we have both just about forgotten what her natural hair color is!

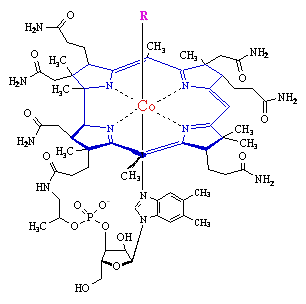
Chemists have synthesized some extraordinarily complex molecules. The synthesis of Vitamin B12 in 1972 (structure below) took 11 years, required more than 90 steps (I typically ask you to do 2 - 6 step syntheses!), and involved over 100 people.
Some more recent examples of complex syntheses are taxol, brevitoxin and ionomycin. Remember, not only were these complex structures assembled from very small constituent parts, the stereochemistry at EACH center had to be maintained! Why are these very complex compounds synthesized? Usually for drug purposes. Taxol, for example, is apparently a promising anti-tumor agent, but can only be isolated in microscopic quantities from its natural source, the bark of the relatively rare and slow-growing pacific yew tree.
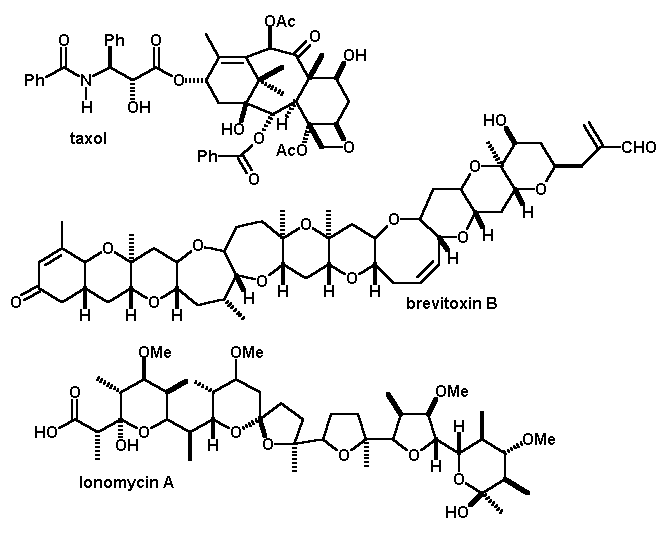
It has been claimed that chemists can now synthesize ANY structure that might be proposed, and that synthesis is a dead research area. I do not agree. Syntheses of compounds such as those shown here require extraordinary commitment of manpower, better and more efficient synthetic methods will always be required!