
O-Chem in Real Life: Appreciating the Reactivity of a Carbon Anion
Carbon anions in which the carbon is sp3 hybridized are essentially impossible to make under normal laboratory solution conditions, the energy of a pair of electrons in a localized sp3 hybrid A.O. on a carbon atom is simply too high. We have already seen the acetylide anion, which is a carbon anion that can be made with a sodium counterion, but in this case the energy of the non-bonding pair of electrons is decreased considerably because they are in a sp hybridized orbital.

To get a pair of non-bonding electrons on a carbon atom, especially in an sp3 hybrid orbital, we need some other way of decreasing the energy of the electrons. The trick that is used is the obvious simple one, just put them into a bond! Not just any bond, though, it has to be a weak bond to keep the electrons reactive enough, and preferably a bond that polarizes the electrons towards the carbon atom so that it can use them in reactions. This is the rationale behind organic metallic reagents.
There is still a bit of debate about the nature of the carbon-lithium bond in organolithiums, exactly how covalent or ionic it is. It is obviously very polar, and polarized with the electrons localized towards the carbon atom, but it is more covalent than an equivalent carbon-sodium or carbon-potassium bond, for example. Electronegativity decreases going down the periodic table AND atomic size increases. Both of these effects make the carbon-metal bond more ionic, due to the larger electronegativity difference and poorer orbital overlap as the metal atom gets significantly larger than the carbon atom. Consequently, organosodiums and potassiums are so ionic that their structures localize the electrons to look more like a pair of non-bonding electrons on the carbon atom, they are extremely reactive, and because they are so polar they tend to aggregate and are basically not very useful.
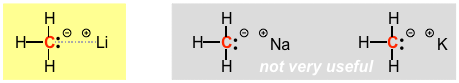
Organolithiums, however, are "just right" in that the bond is strong enough so that the electrons are lowered enough so that the structures can be prepared and handled, but weak and covalent enough so that they REALLY want to get into a stronger bond with an element such as hydrogen, i.e. they are VERY strong and VERY useful Bronsted bases. (NOTE that the structure shown above on the yellow background is very unconventional in that it attempts to show the mixed ionic and covalent character of the bond in the dashed grey line). Even though they are not truly carbon anions, they are much stronger bases than equivalent nitrogen and oxygen anions.
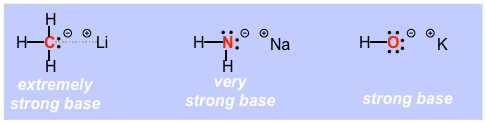
So, it is very easy to say "of course a carbon base is stronger than a nitrogen base etc due to electronegativity", but it is difficult to appreciate HOW much stronger, we even lose track of the real meaning of pKa, so here is a different illustration of base strength via chemical reactivity. If you draw a concentrated solution of an alkyl lithium into a syringe, t-butyl lithium is a very good example, and by solution I mean solution, alkyl lithiums are usually used as solutions in alkane solvents such as hexane, and then squirt the solution out of the syringe you will then have what is effectively a small flame thrower as the reagent rips protons out of any water in the atmosphere in a reaction so exothermic that it sets fire to the alkane solvent. This is the power of a pair of non-bonding electrons on a sp3 hybridized carbon atom even when they are still in a weak bond!
I thought that a picture of this might be nice, but when you do a Google search you only find the one below, which is the same thing, not with an alkyl lithium but an alkyl zinc, same kind of thing, but I have to admit that the zinc one is more dramatic than you get with the alkyl lithium.

Playing with alkyl lithiums like this is what organic chemistry graduate students do late at night when they are running very long reactions and they get bored! Of course this is dangerous, and you are not supposed to do this, etc...
Actually, alkyl lithiums really can be dangerous, and nobody should use them unless they REALLY know what they are doing, and should not work alone with them. There was a very tragic incident a few tears ago at UCLA in which someone working in a research lab had a very bad accident when using t-butyl lithium and unfortunately died. So, carbon bases are very reactive and need to be taken seriously