O-Chem in Real Life: Substitution Reactions and Energy in Real Life
The main guiding principle we are talking about in class is that the energies of the electrons are perhaps the most important property in determining the structure, shapes and reactions of organic molecules. In fact, this is almost always true.
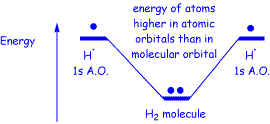
We also talk about the fact that electrons want to get into bonds if they can, because by doing so they lower their energy. Furthermore, we have said that as far as our organic chemistry classes are concerned, we can assume that just about all electrons that are NOT in bonds are HIGHER IN ENERGY than just about all electrons that ARE IN BONDS. And this is also mostly true, the classical example being formation of the hydrogen molecule from two hydrogen atoms.

If this were always the case, then we would expect a chemical process that converts non-bonded electrons into bonded electrons to give off energy (be exothermic), and a process that converted bonded electrons into non-bonded electrons would require energy to be added, and would be endothermic. We could also go further, and say that processes that are very exothermic (give off a lot of energy) should presumably be associated with formation of either a lot of bonds, or by the conversion of weak bonds (which contain high energy electrons) into strong bonds (which contain low energy electrons), and again, this will generally be true in the organic reactions that we will study.

However, this is NOT true for one crucially important system, in fact the system upon which most energy conversion takes place in living things, i.e in the reactions of ATP. ATP (shown below) is often described as the energy currency for the myriad chemical reactions that occur in living things. The name is an abbreviation of Adenosine TriPhosphate. The adenosine part of shown in red (the upper aromatic part is adenine) and the triphosphate is in blue. What follows is an extremely abbreviated summary of the important role of ATP in living organisms. To learn more you will really have to take a biochemistry course.
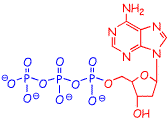
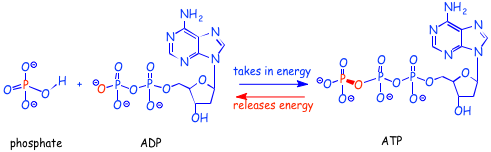
When energy is extracted from decomposition of food, a reasonable part of that energy is used to make ATP from ADP (below), by the formation of a new oxygen-phosphorus bond, as shown below. It requires energy to convert ADP to ATP, and once ATP is formed, that energy is "stored" (in the form of chemical energy) in the ATP. You probably first heard about chemical energy in grade school. Now you know that chemical energy is "stored" in the form of higher energy electrons, the thing that we have been talking so much about in class. So, when ADP is converted into ATP, the energy of some electrons are raised.
This reaction is SUBSTITUTION, but the mechanism is not a simple SN2, as shown below.
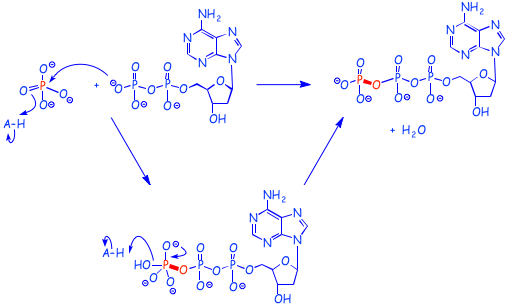
Conversely, when ATP is converted into ADP, energy is released. The scheme above is horribly simplistic, since the processes above are usually incorporated into other reactions involving enzyme catalysis, and protonation and deprotonation steps that are not shown, but this will do for now.
The net effect of going from phosphate + ADP to ATP is illustrated by the bonds in red above. In essence, one kind of oxygen-phosphorous bond is converted into another kind of oxygen-phosphorous bond. The number of electrons in bonds and the types of bonds they are in are essentially unchanged. So how does the electronic energy change enough that this is such a useful energy conversion reaction in nature?
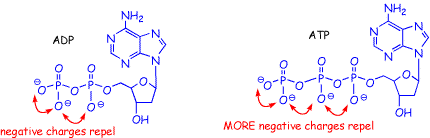
Well, there are still differences in the energies of the electrons on both sides of the equations, it is just that in this case the energy differences are mainly determined by factors other than whether electrons are in bonds or not. There are several reasons that are usually given to account for the energy change. The first is that in ATP there are four negatively charged oxygens. These excess negative charges raise the energies of all of the electrons, especially those on the oxygens as a result of the charge imbalance (there are not enough positively charge protons to stabilize or lower the energies of the electrons). On ADP, there are three negatively charged oxygens, so the electron imbalance is less on ADP and the electron energy is lower.
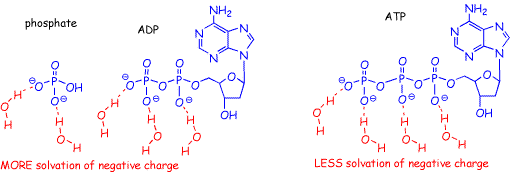
The second reason is a consequence of the fact that this reaction occurs in water. The H-O bonds in water are highly polarized, with a small effective positive charge on each H. The hydrogen atoms on water are thus capable of stabilizing (lowering the energy) of electrons in solvated molecules, particularly those on atoms with formal negative charges (anions). Because there are more anions in the ADP/phosphate combination, this contributes to a lowering of energy on this side of the equation.
Finally, we are learning that resonance stabilizes non-bonding electrons, since in effect this allows them to "see" more nuclei. Formation of ATP from ADP removes the possibility of resonance stabilization of electrons on one of the oxygens, which also contributes to lowering the overall energy of the electrons on the ADP side of the equation.
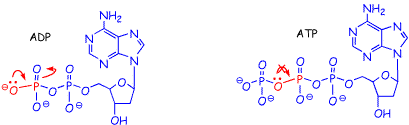
Please remember that the above description is a ludicrously simplified description of the role and functions of ATP, but I did think it worth visiting this important issue at this time, and it shows that real life chemistry is often more complex that we talk about in class!