Organic Chemistry in "Real Life"
This week's "Real Life" page is borrowed from last semester, since we are returning to a discussion of molecular orbitals. In first semester organic we said that molecular orbitals told us "where the electrons are" in a molecule. You will recall that combination of two atomic orbitals generates two molecular orbitals, one bonding and one anti-bonding. As we will see in more detail in the next couple of weeks, the bonding molecular orbitals are where the electrons are. But what about those apparantly useless antibonding M.O.'s? Why do we care about them? In the next couple of weeks we will need to know as much about the anti-bonding M.O.'s as the bonding M.O.'s to understand certain reactions. Remember, just because there are no electrons in the anti-bonding M.O.'s, it doesn't mean that they aren't "there" to accept electrons. A hydrogen atom "has" 2s and 2p atomic orbitals even if it doesn't usually have any electrons in them. Soon, we will be making bonds by "transferring" electrons from one molecule to another. Electrons can not be accepted by a molecule into a bonding M.O., it is full, so the electrons have to go (temporarily) into antibonding M.O.'s. If we don't understand the nature (and the phase and nodal patterns) of the antibonding M.O.'s we will not be able to predict the bonding correctly.
There is another area in which the antibonding orbitals play an important role, i.e. in determining the colors of molecules. Why are things colored? To understand this you need to know something about light.
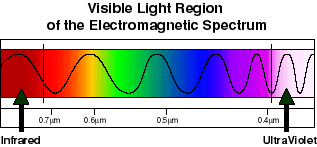
Light that we can see is an example of electromagnetic radiation, more specifically it represents that part of the electromagnetic spectrum that our eys are sensitive to. The difference between red light and blue light (for example) is the frequency of the electromagnetic oscillations. Red light has low frequency or (relatively) long wavelength oscillations, blue has higher frequency or (relatively) short wavelength oscillations. Red represents the lowest frequency (longest wavelength) that we can see. Light that has lower frequency (longer wavelength) oscillations than red light is infrared. Blue represents the highest frequency (shortest wavelength) we can see. Light that has higher frequency (shorter wavelength) oscillations than blue light is ultraviolet. Green light is in the middle of the range of frequencies (wavelengths) that we can see. The wavelength of red light is roughly 0.7 mm, or 700 nanometers. Blue light is roughly 0.4 mm, or 400 nm. White light is a mixture of blue, green, red etc light. White light can be split into its constituent colors by passing it through a prism. The energy of light is directly proportional to its frequency. Low frequency (e.g. red) light has a (relatively) low energy. High frequency (e.g. blue) light has a (relatively) high energy.
|
|
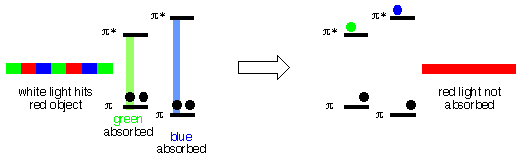
When white light (the mixture of colors) from a light bulb, the sun etc., shines on an object that is red, the red part of the white light is reflected off the object to your eyes. The blue and the green parts do not reflect, they are "absorbed" by the object. A blue object reflects only blue light (not red or green), a green object absorbs red and blue, etc. Other colors are "made" by mixing the primary colors in different proportions etc. A black object absorbs all wavelengths of light that hits it, a white object reflects all wavelengths of light that hits it.

So, how is the light absorbed? The light energy has to go somewhere, it has to be "used". It turns out that the energy of light in the visible region can be close to the energy difference between bonding and antibonding Molecular Orbitals in molecules. When light is absorbed, the energy is used to move an electron from a low energy bonding (usually p) M.O. to a higher energy antibonding (p*) M.O. If the energy difference between a bonding p M.O. and its corresponding p* M.O. is equal to the energy of (for example) green light, then the green light can be absorbed. If the energy difference between a bonding p M.O. and its corresponding p* M.O. is equal to the energy of (for example) blue light, then the blue light can be absorbed. In the example below, there is no bonding p M.O./p* M.O. pair for which the energy difference is the same as that of red light, so the red light is not absorbed.
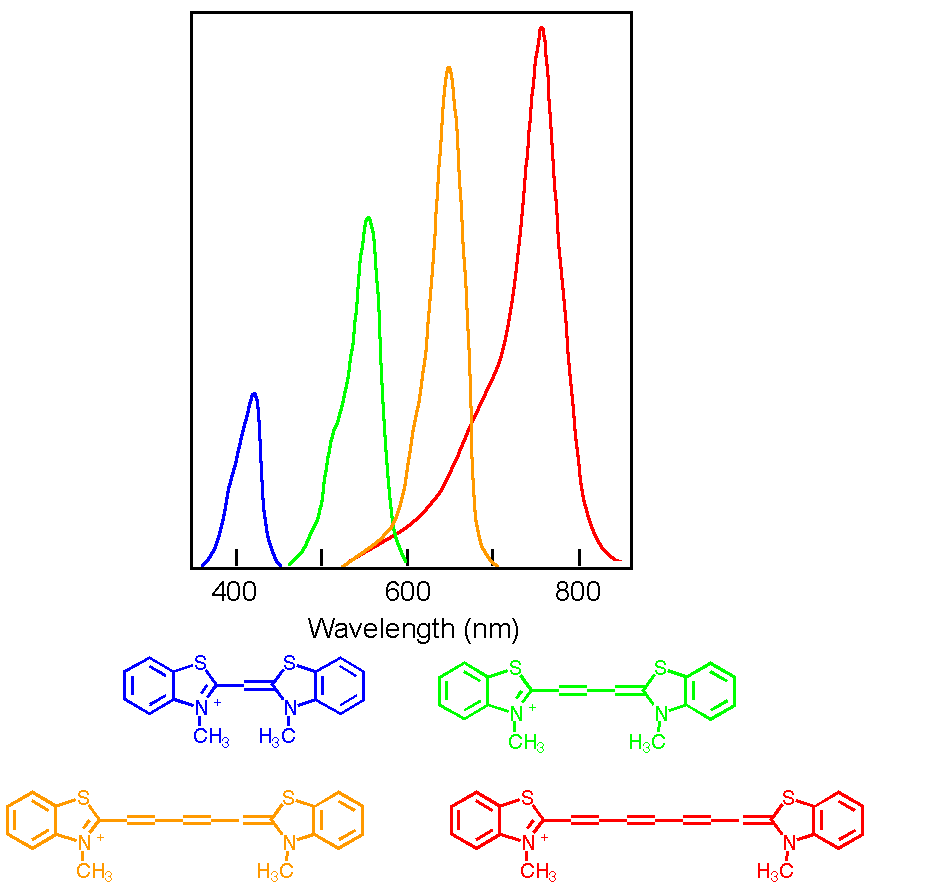
Chemists can control the energy difference between the p M.O. and p* M.O.'s to control the colors of the light absorbed. This is how dye chemistry works! In general, the energy difference gets smaller the bigger the molecule. Here are some dye molecules that are used in photographic film. The different dyes make the film sensitive to different colors of light, blue, green, orange, red etc. The figure below shows how much the different dyes absorb the different wavelengths of light.
What happens to the electrons once they are "promoted" into the antibonding M.O.'s. As you can imagine, they are very reactive, being so high in energy. The energy of these electrons can be used to initiate many useful reactions, such as those in imaging such as photography or xerography. If the light that hits the molecules is sunlight then they can also be used to initiate chemical reactions that can store the light energy in solar conversion schemes. If the light is sunlight and the dyes are in plants, then the light absorption can be used to initiate the photosynthesis process. So you see, the antibonding orbitals are used in the most important chemical processes, those responsible for life itsself :)