O-Chem in Real Life: A 2 + 2 Cycloaddition
In class we learned that (concerted) 2 + 2 cycloaddition reactions are forbidden. Indeed, such concerted reactions are essentially never observed. Never, that is, except in the presence of light. Our textbook tells us that concerted pericyclic reactions that are forbidden, are often allowed in the presence of light. Pericyclic reactions that are thermally forbidden, are described as being photochemically allowed since the light promotes an electron from the HOMO to the LUMO, which has opposite symmetry properties, thus allowing bonding interactions in the transition states.
We do not cover photochemical reactions in our course for several reasons. First, we don't have time to cover all topics, so this is one that I choose to drop. Second, I don't believe the explanation of photochemical reactions in the textbook! It is true that 2 + 2 cycloadditions are observed photochemically, whereas they are never observed in non-photochemical reactions, however, most of these are not concerted, and thus the F.M.O. and Aromatic Transition State rules don't apply. In fact, photochemists are still searching for unambiguous examples of photochemical peericyclic reactions!
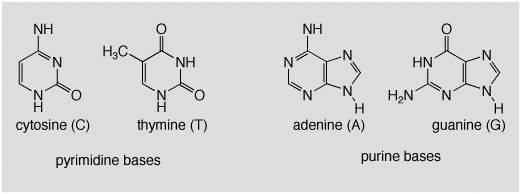
One 2 + 2 photochemical cycloaddition that may be a true pericyclic reaction is the dimerization of thymine in DNA. Thymine is one of the four common organic bases found in DNA. The structures of the bases are shown below.

As we all know, the ordering of these four bases along the DNA backbone results in the genetic coding required for replication of proteins etc. The bases are arranged as parallel hydrogen bonded pairs on the indise of the well-known double helix DNA structure, shown below.
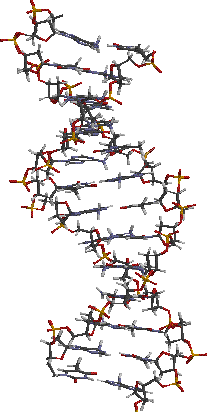
Exposure of DNA to light can induce photochemical reactions of the thymine bases, and when they are in adjacent positions on the DNA backbone, a 2 + 2 cyclodimer can form, shown below.
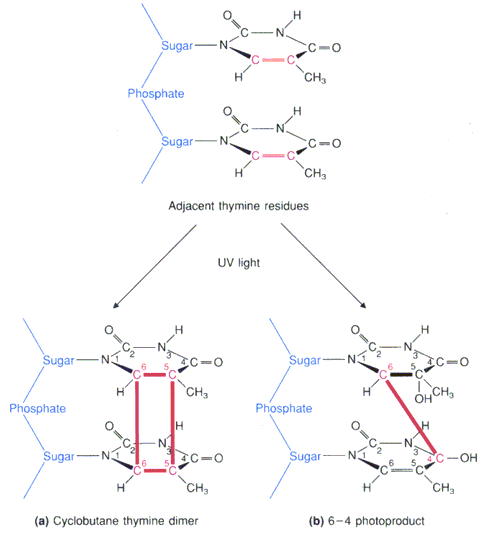
Formation of such a dimer introduces a lesion into the DNA chain. Such thymine-thymine lesions have been linked to the development of cancerous cells, and in particular have been related to development of skin cancer.
Many organisms have an enzyme that binds to such Thymine-Thymine lesions, and with further exposure to light cleaves the cyclobutane ring and repairs the lesion. This enzyme is not present in humans. Humans do have other enzymes that can excise (cut out) the dimerized bases and replace them with new bases, however, it is still a good idea to keep using the sunblock.