O-Chem in Real Life: Polycyclic Aromatic Hydrocarbons (PAH's)
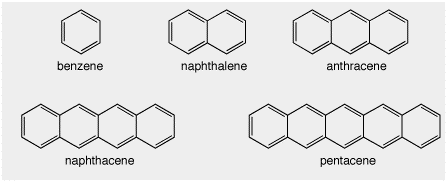
Here are some aromatic compounds. Benzene we know, of course. The other, larger, structures, are also aromatic based on Huckels rule, i.e. (4n + 2) electrons in a cyclic loop.

PAH's are often termed "persistent", or "bioaccumulative". This means that they are difficult to metabolize and/or excrete, and thus tend to accumulate in the body. PAH's are usually pretty toxic too! Exposure to PAH's usually occurs by breathing air contaminated by wild fires or coal tar, or by eating foods that have been grilled. Other sources include industrial processes, transportation, energy production and use, smoking tobacco, and disposal activities such as open trash burning. Not good, huh?
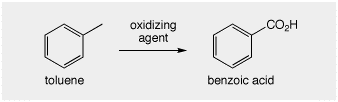
To understand some of the problem, let's compare benzene and toluene. Benzene is considered carcinogenic, whereas toluene is much less toxic. We would oxidize toluene into benzoic acid in the lab (or on midterm 3!) using permanganate. Enzyamtic conversion of toluene to benzoic in the liver renders it water soluble and excretable via the kidneys. Corresponding oxidation of the pure aromatic structure benzene is not possible.
| Why does Dr. Gould care about the carcinogenicity of benzene? |
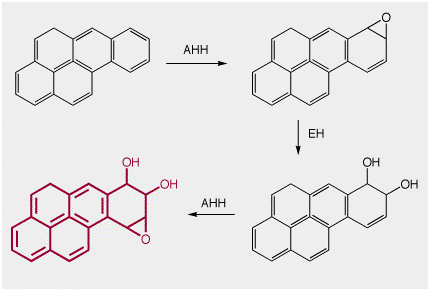
With increasing number of rings, the aromatic rings of PAH's do become more reactive. This is because the large the number of electrons in the aromatic system, the higher the energy of the HOMO. At its root, all oxidations can be understood as starting by removal of a single electron, and this is easier the higher the energy of the electrons in the HOMO. The multi-ring Benzopyrene (see above) was one of the PAH's earliest identified as a carcinogen. It was found by extraction of TWO TONS(!!) of gas-works pitch, and is now recognised as the first industrially generated chemical carcinogen
Actually, it is perhaps better defined as an "indirect" carconogen. This large ring system IS oxidized in the liver (the liver is just doing it's job, trying to oxidized everything that comes its way!) using the enzymes Arylhydrocarbon Hydroxylase (AHH) and Epoxide Hydratase (EH), to form the maroon epoxy-diol below. This epoxy-diol can intercalate between the bases in DNA, and the epoxide can act as an electrophile to one of the amines of the DNA purine bases, forming a DNA adduct that blocks normal DNA replication.
Of course, tomorrow you may be hit by a bus, so why worry!