
O-Chem in Real Life: Nitrogen
Carbon, hydrogen, oxygen are all abundant elements on earth that form strong covalent bonds, which is why they are commonly found in organic and biomolecules. Another common element is nitrogen, but there is also loads of nitrogen in the air (the air is 80% nitrogen), which might make us ask why, and wouldn't it be better if organisms were abel to access that nitrogen to make useful organic structures out of.
There is so much nitrogen in the air because it can form a very strong N-N triple bond. Carbon can't form a strong C-C quadruple bond, can it? (you know why, think about it....) Oxygen can;t form a string O-O double bond, can it? (you were supposed to learn that in gen chem, it is a molecular orbital argument). But nitrogen can form a very strong triple bond and then it becomes very stable (it is hard to break a triple bond between atoms of the same electronegativity). Nitrogen gas is also a very light diatomic molecule and so it floats up away from the earth into the atmosphere!
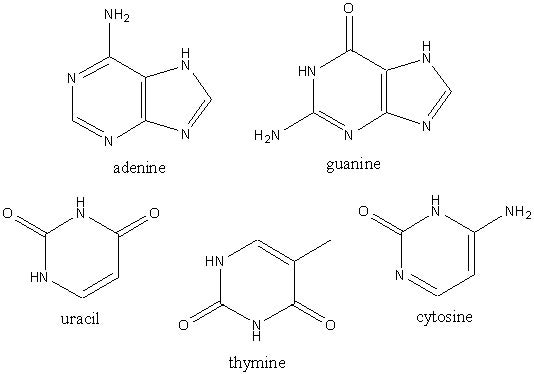
So now there is a problem, how to plants and animals use the nitrogen to make useful stuff? Nitrogen is actually pretty important to plants and animals, for example in amino acids (below left) and the DNA bases (below right).


Neither plants or animals can actually get molecular nitrogen from the air, however, bacteria can, in a process called nitrogen fixation.
Nitrogen fixation is the process whereby molecular N2 is converted into a form of nitrogen that plants can use specifically the ammonium ion, NH4+ or nitrogen dioxide, NO2. This conversion from essentially inert N2 into more reactive forms of nitrogen is essential to life, and it is a pretty neat trick that bacteria can do it without using any highly reactive reagents or extreme conditions. This is another area where nature beats us badly.
Chemists can also fix nitrogen by reducing it to ammonia, NH3, is a process that is roughy similar to the catalytic reductions of alkenes to alkanes and alkynes to alkenes using H2 and metal catalysts such as Pd/C and the Lindlar catalyst. It is called the Haber process and it requires very high temperatures (around 400 - 500 degrees C) and high pressures of hydrogen gas (around 200 atmospheres). And so, yes, we can do what the bacteria do, but we do it in a very inefficient way that uses a lot more energy than nature.