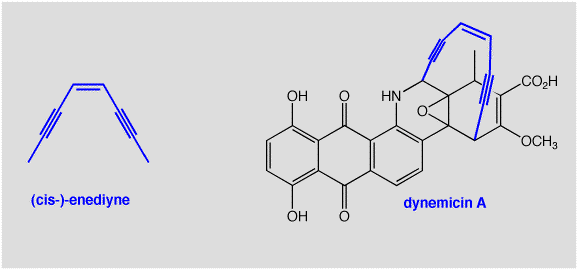
O-Chem in Real Life: A Peculiar Alkyne
An enediyne is a conjugated system consisting of consecutive triple, double and triple bonds. This unusual sequence of functional groups is actually found in several natural systems. In particular, the compound dynemicin A has been found in certain molds.

Why the interest in this unusual structure from, of all sources, a mold? One reason is that dynemicin A has been found to inhibit the growth of cancer cells. The origin of its activity has been traced to the enediyne part of the molecule. The enediyne has been found to undergo a very unexpected (and very cool!) cyclization reaction (below). This reaction is another reason that enediynes have been studied so much recently. The reaction looks very strange, since the product is a diradical, with TWO unpaired electrons! In essence, it transfers two electrons from bonding to non-bonding orbitals, and we usually say that is a very bad thing to do, or at least it should cost quite a lot of energy.
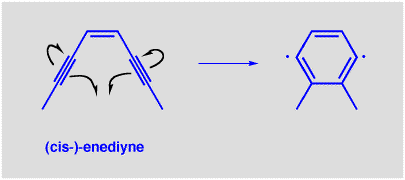
The energy cost associated with formation of the diradical must be offset by some energy gain, otherwise this reaction would never go, and it is. The product is also a benzene ring. Benzene is also a conjugated system, but cyclic (aromatic) conjugated systems such as these have a special stability associated with them. We will be studying aromatic systems soon, but for now we just have to accept that a reaction that forms an aromatic system is usually associated with a (sometimes substantial) decrease in energy. In dynemicin A, you can see another source of driving force for the cyclization reaction, since the alkynes are not quite linear due to ring strain. This strain is relieved as a result of the cyclization.
Still, the biradical that is formed is pretty reactive, and it really wants to get those non-bonding electrons into a bond if it can. Here is where the anti-cancer activity comes in. It is thought that dynemicin A can interact with DNA at the cell level, and that the diradical can form bonds between the two chains of double-stranded DNA. This cross-linking of the strands can prevent replication, and hence interfere with cell growth.
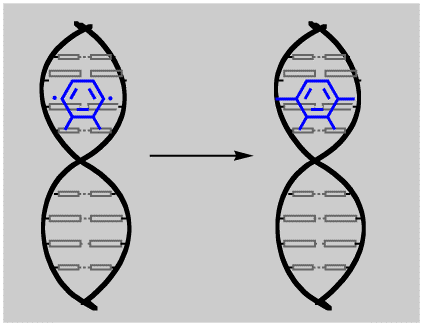
Of course, it is not obvious why this cross-linking reaction should discriminate between cancer cells and healthy cells, and in fact, it doesn't! Chemists are trying to synthesize analogous molecules that contain the enediyne functionality that might be less toxic, but still useful as anti-cancer drugs. I am not sure how they hope to introduce thus discrimination between cells, I suspect that they are just being hopeful, which is why I could never do this kind of research.