
O-Chem in Real Life: The Alcohol That Some People Drink
Alcohol (ethanol) remains the drug of choice for most Americans. There is some chemistry related to the recreational use of alcohol that we should be able to understand.
Alcohol is water miscible, which means that it is rapidly absorbed by the body, and that is also rapidly spreads throughout the body. This means that it also easily crosses the placenta, thus entering the fetal circulation system, which is not good. Alcohol has been shown to affect a large variety of body functions and pathological conditions related to most body organs have been demonstrated. However, it easily crosses the blood/brain barrier, and it is as a consequence of entry into the central nervous system that most of the effects we are aware of probably arise. So, basically, when you drink, you can think about it as if your brain gets immersed in ethanol, nice, huh?

90% of the alcohol that is drunk is metabolized in the liver. The most important enzyme involved in ethanol metabolism is alcohol dehydrogenase, which in the presence of the co-enzyme NAD, converts ethanol into acetaldehyde. This is obviously an oxidation reaction, and one that converts a primary alcohol into an aldehyde. To do that in the lab, we would have to use PCC, which contains no water. Our bodies manage to do this despite the fact that we are mostly water! The enzyme is a claver thing. This reaction takes place only in the liver and its rate is completely unaffected by the consumption of caffeine, or immersion ion cold water etc., these things will not prevent you from getting drunk! Acetaldehyde is toxic, but is in-turn further oxidized to acetate which can be used in other metabolic processes. So, alcohol is a foodstuff of sorts, just not a particularly good one. Ethanol is produced in our bodies naturally, which is presumably why we have alcohol dehydrogenase in the first place.
Several of you will have heard me say this before. If you want to get good a tennis, you practice playing tennis, if you want to get good at drinking a lot of beer and not falling over, you drink a lot of beer, if you want to get good at doing organic chemistry tests you do a lot of organic chemistry problems. The beer thing really does work. In response to exposure to increased ethanol, most human bodies build up the steady-state concentration of alcohol dehydrogenase, and when this happens it takes more alcohol to get you drunk. The well-known increased alcohol tolerance that alcoholics achieve is almost certainly related to adaptations in the central nervous system, and this build-up of dehydrogenaze enzymes in the liver.
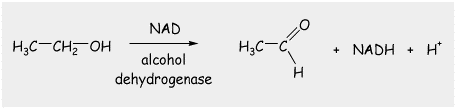
Alcohol that is not metabolized is excreted in urine, or by breathing. This latter phenomenon is the basis for the "breathalyzer" test. When I lived in the UK there was a simple test used by police to determine whether or not you had been drinking, that used the same chemistry of the dichromate ion oxidation we learn in second semester organic. Yellow crystalline dichromate oxidizes ethanol and is in turn reduced to the chromium III ion, which is green (actually, it is blue, but a mixture of blue and residual yellow looks green). When pulled over you were asked to blow into a bag through a tube containing the dichromate. If the crystals turned green you were in trouble!.
Of course, the breathalyzer test described above isn't very selective, since dichromate will oxidize any alcohol (and other organic compounds), such as methanol. It turns out that the alcohol dehydrogenase isn't very selective either, it also oxidizes many alcohols. When methanol (wood alcohol) is oxidized, it forms formaldehyde. Formaldehyde is extremely toxic in our bodies, and thus methanol is very poisonous. So although a breathalyzer test would not be able to distinguish whether you had consumed ethanol or methanol, there is another breath test that does, i.e. whether you are actually still breathing or not after consuming the alcohol! Unfortunately, death due to inadvertant consumption of methanol in the place of ethanol is not uncommon.
Finally, here is a rough guide to the effects of various blood alcohol levels on your otherwise normal behavior. Try to avoid a blood alcohol concentration of greater than 0.45%.
| % Blood Alcohol | Effect | Clinical Symptoms |
| 0.01 - 0.03 | Sobriety | Behavior nearly normal. Slight changes detectable in reaction tests and information processing of two or more stimuli |
| 0.04 - 0.08 | Euphoria | Euphoria, sociability, talkativeness. Decrease of inhibitions. Diminution of attention. Decrease in judgment and control. Decrease in finer performance |
| 0.09 - 0.15 | Excitement | Emotional instability. Loss of inhibitions. Loss of critical judgment. Loss of muscular coordination |
| 0.15 - 0.30 | Confusion | Disorientation. Exaggerated emotional states. Disturbance of sensation and perception of color, form, motion, dimensions. Further loss of muscular coordination, staggering gait, slurred speech |
| 0.30 - 0.45 | Coma | Markedly decreased response to stimuli. Marked loss of muscular coordination, inability to stand or walk. Approaching paralysis. Impaired consciousness |
| > 0.45 | Death |