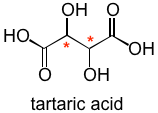
Tartaric Acid
Tartaric acid is of high importance historically in chemistry. As you can see, it has two chiral/asymmetric centers (*).
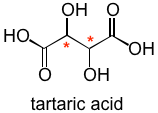
With two chiral centers, multiple stereoisomers are possible, but it is the (R)(R) and the (S)(S) stereoisomers that relevant to this story.

These 2 structures are obviously chiral, and indeed, they are also obviously a pair of enantiomers. Because they are chiral they are also optically active, meaning that they will rotate polarization of plane polarized light.
But in the 1800's there was a mystery about tartaric acid. At the time, stereochemistry was not really understood, but it was known that tartaric acid that as isolated from plants was optically active. Tartaric acid was also formed in wine fermentation, but the tartaric acid from wine was not optically active. The two tartaric acids were known to have the same structure and undergo the same reactions, so what was going on?
Louis Pasteur was interested in this and was also interested in crystals, and so he looked very carefully at the crystals of the non-optically active tartaric acid (actually, salts of the acids, but that is a detail) and found that there were two kinds of crystal, one a mirror image of the other.
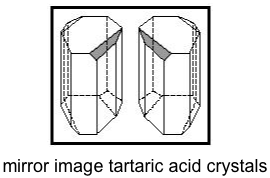
When Pasteur painstakingly separated the to kinds of crystal, he found that each were optically active and that each rotated polarized light in the OPPOSITE direction. Pasteur has discovered optical isomerism, and from there the jump to understanding stereochemistry as due to the arrangement of atoms at chiral/asymmetric centers was short.
The tartaric acid from wine was a racemic mixture, and that from plants was a single enantiomer. It turns out that the one that plants make is the (R) (R) enantiomer. You should be AMAZED that the mirror image relationship between the molecular structure can be reproduced in the macroscopic mirror image relationship in the shape of the crystals!